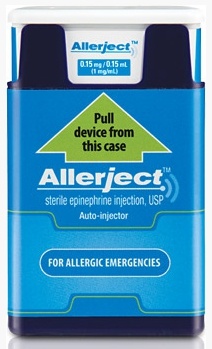
Parents and caregivers are being advised to return the recalled products directly to their pharmacy for a replacement product with the same dosage.
The products (Allerject 0.15 mg / 0.15 mL auto-injector, lots 2857508 and 2857505, DIN 02382059) started being distributed on June 1, 2015.
Parents and caregivers are being advised to return the recalled products directly to their pharmacy for a replacement product with the same dosage.
For more information about the recall click HERE for the Health Canada notice.



 WFD Presents 2024 Report to City Council
WFD Presents 2024 Report to City Council
 Council Awarded Organizations with Community Grants
Council Awarded Organizations with Community Grants
 Battle of the Badges this Weekend
Battle of the Badges this Weekend
 OPP Reminding Drivers to Stay Focused
OPP Reminding Drivers to Stay Focused
 Reduced Load Campaign Continues in Oxford
Reduced Load Campaign Continues in Oxford
 Five Impaired Drivers Charged in Oxford
Five Impaired Drivers Charged in Oxford
 UPDATE: Missing Girls Located
UPDATE: Missing Girls Located
 SWPH Reports 18 New Measles Cases
SWPH Reports 18 New Measles Cases
 Brant Parks Closed Due to Flooding
Brant Parks Closed Due to Flooding
 Sip and Sample is Back
Sip and Sample is Back
 New Minimum Wage vs. Living Wage
New Minimum Wage vs. Living Wage
 WITAAR Announces 2025 Charity of Choice
WITAAR Announces 2025 Charity of Choice
 Online Spring Auction Underway for OCAR
Online Spring Auction Underway for OCAR
 Measles Exposure Reported at TDMH
Measles Exposure Reported at TDMH
 WPS Investigating Harding Court House Fire
WPS Investigating Harding Court House Fire
 UPDATE: Rain Warnings End
UPDATE: Rain Warnings End
 Enrollment Cuts Coming at Fanshawe College
Enrollment Cuts Coming at Fanshawe College
 Charges Laid in Paris Assault
Charges Laid in Paris Assault
 BBBS Oxford Welcomes MEGABike to Tillsonburg
BBBS Oxford Welcomes MEGABike to Tillsonburg
 CASS Presents: The Wedding Singer
CASS Presents: The Wedding Singer



Comments
Add a comment